The database
The combination of our ultra-fine bubble generating system and advanced bubble measurement technology enabled us to obtain a wide range of information concerning which materials are easier to “bubblize” and which production protocol should be applied. We have tested hundreds of materials ranging from distilled water to glucose, salt, protein, lipid included solutions. Also, various gas type such as hydrogen, argon, oxygen, helium and others, have each shown different biophysical bubble behavior. Our strength lies on this huge multi-parameter database and know-how obtained through intensive experiments conducted at our lab. Built on this platform, our “core” technology can be apply to futuristic pharmaceutical products or for re-innovating already approved drugs, transforming them into theranostic functioning agents. Because no additional chemical revision is needed in this technology, careful strategic planning could lead to early FDA approval.
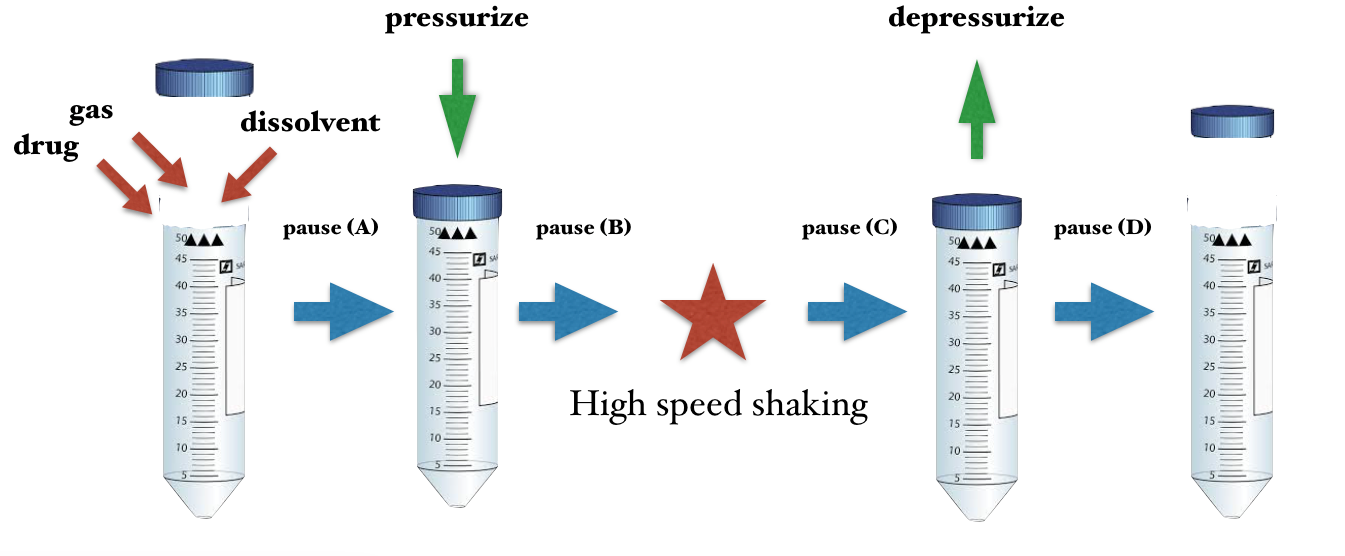
The basic protocol of generating ultra-fine bubbles consists of many conditions, materials and procedures. The combination of these parameters are infinitive but have to some extent, a constant “pattern” that can be directly applied to similar pharmaceutical products.